All MMK GC qualities certified for laser and inkjet coding, perfect for 2D matrix codes
31.12.2015 - Virgin Fibre Cartonboard
The ordered implementation of the EU Falsified Medicines Directive will in future oblige every pharmaceutical packaging to carry its own unique identifier. Mayr-Melnhof Karton can already meet the increased quality demands placed on cartonboard: an external institute confirms the excellent laser and inkjet codability of all MMK virgin fibre qualities.
New EU Directive requires two safety features
In February 2016, the European Commission published the final version of the delegated acts which will define the specific details of Directive 2011/62/EU (Falsified Medicines Directive). The Directive serves to make the fight against counterfeit drugs more effective and therefore aims to protect patients. The regulation provides for safety features which can be divided into two groups:
- Anti-tampering devices such as glue dots, adhesive seals and perforated tear strips will in future allow inspection to see if a carton has been manipulated.
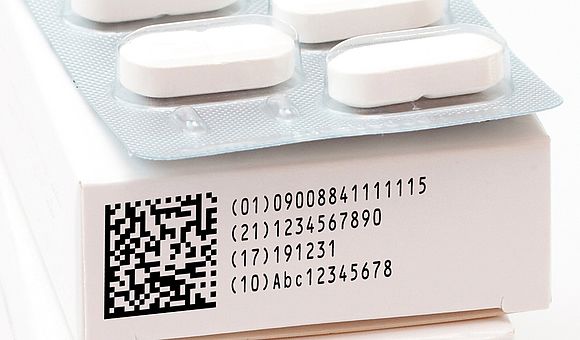
- The unique identifier on every pharmaceutical packaging – the 2D matrix code – will serve to show if a drug is genuine and to identify the individual packaging. These codes can supply additional information and thereby allow seamless product traceability as well as reliable counterfeit protection. The fundamental requirement here is the optimum codability of the cartonboard.
MMK‘s GC qualities meet requirements in full
MMK‘s R&D department has already spent several years working to meet the future requirements in full. All laboratories in our virgin fibre cartonboard mills have special equipment to test the coding and its readability under real conditions.
When a product is coded with a laser, the surface layer is quickly and precisely vaporised or engraved, thereby achieving a product coding that cannot be rubbed off. With individual inkjet coding, the 2D matrix code is printed on the folding carton directly in the pharmaceutical packaging plant. The challenge
with these inkjet codes is that they have to be dry and wipe resistant within 0.3 seconds in order to be correctly readable in accordance with the FFPI guideline (Forschungsgemeinschaft Faltschachteln für die Pharmaindustrie, engl.: Research Group Folding Boxes for the Pharmaceutical Industry) and the criteria contained in ISO standard 15415. This requires optimised coating of the cartonboard quality. The optimum coding properties of our GC qualities, both for laser and inkjet coding, have now been confirmed by PTS (Papiertechnische Stiftung): all MMK virgin fibre qualities as well as the WLC products, Multicolor Mirabell™ (GD2), Starcolor Mirabell™ (GD2) and Supra™ (GT2) are excellently suited for the use of 2D matrix codes and are therefore first choice for pharmaceutical companies.
Scope and timetable for implementation
It will be compulsory to implement the Falsified Medicines Directive by the beginning of 2019: it will comprise all prescription drugs sold in the European Union. Exceptions are shown on the so-called white list. The new regulation do not yet apply to non-prescription, OTC drugs: the safety features are not to be compulsory for OTC products. Drugs where there is a high risk of counterfeiting form an exception to the above (black list).
Whether a drug requires a prescription is normally decided at a national level and can be different in individual member states. This means it is possible that one and the same drug will have to carry the safety features in one member state but not in another.
Continuous investment in research and development together with deployment of the latest technology form the basis for us to take account of market trends and imminent legislation at an early stage. It is only thanks to this approach that MMK is already in a position today to meet the rising demands of tomorrow.